In commercial and institutional buildings, cooling systems are not exactly top of mind when it comes to conservation and the use of “green” technologies. Yet the potential for significant cost and water savings and reduced environmental impact exists. With more-stringent effluent and chemical-handling regulations, increasing drought conditions, and fluctuating chemical prices, facility owners increasingly are looking to cooling towers — more specifically, the treatment of cooling water — to meet the objectives of sustainable cooling: more efficient equipment performance, saving makeup and discharged water, and reducing or eliminating the use of chemicals.
To effectively manage a cooling tower, one must understand it in terms of:
-
Water use
Exactly how much water does the system use on a yearly basis?
-
The type and amount of chemicals used
TRADITIONAL METHODS
Are the chemicals potentially hazardous to workers? What is the actual cost of use? Are there more effective alternatives? How volatile are prices?
-
Discharge regulations
What discharge limits are imposed by the local publicly owned treatment works (POTW)? Is the facility in violation?
-
Environmental impact
Are harmful chemicals being discharged into the environment?
-
Operational efficiency
Poor water treatment can adversely affect tower efficiency (e.g., scale formation, biofouling) and impact heat transfer.
-
Water-reuse options
Can discharge water be used for other purposes (e.g., landscape irrigation, equipment washing, dust control) on site?
-
Worker safety
Is the current treatment technology a health or safety hazard? Does it require moving heavy supplies?
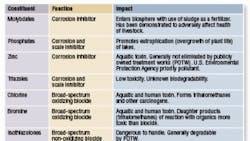
More than 95 percent of the time, chemicals are used to treat cooling water for scale, corrosion, and bacteria. A partial list of commonly used chemicals and their potential issues is given in Table 1.
As dissolved-solids levels increase, so do scale-deposition tendencies. With chemical treatment of cooling water, cycles of concentration are limited by an inability to prevent scale formation. This is attributable to the way in which most chemicals work. They prevent the formation of calcium carbonate by sequestering calcium, preventing crystal growth, or keeping the material in solution. Ultimately, a saturation limit is reached, and the addition of more chemicals becomes ineffective. This is in contrast to non-chemical technologies, which form calcium-carbonate solids and remove them from water, increasing cycles of concentration.
A recent trend in chemical treatment is the use of dry products, which can be used for scale prevention, corrosion protection, and microbial control. Because they usually are concentrated (up to 100-percent active), they are significantly smaller by volume, lighter, and much easier and safer to handle, store, and transport than their liquid equivalents.
With a properly designed treatment program and continuous monitoring, chemicals can be an effective means of maintaining good water quality in a cooling tower. However, facility managers are beginning to seek alternatives because of environmental concerns.
DISCHARGE OF CHEMICALLY TREATED WATER
Most of the chemically treated water discharged from a cooling tower is classified as wastewater. Depending on the nature of the discharge and the route of disposal, regulatory requirements may apply. The four routes of disposal of cooling water are:
-
Holding tanks for transport to a treatment facility.
-
Groundwater discharge.
-
Sewer discharge.
-
Surface-water discharge.
Disposal via holding tanks and groundwater is uncommon and will not be discussed here.
Local and state permits for sewer discharge generally are less stringent than those for surface-water discharge because water goes through a sewage-treatment plant prior to being discharged to the environment. However, most sewage-treatment plants, in an attempt to conserve treatment capacity, prohibit — or at least discourage — the discharge of cooling water. A local POTW should be contacted about its regulations and permit requirements.
Surface-water discharge falls under the National Pollutant Discharge Elimination System (NPDES), authorized by the Clean Water Act, while federal and state laws require permits for surface-water discharge. The NPDES limits flow, conventional pollutants (e.g., biological oxygen demand, pH, total suspended solids, fecal coliform, oil and grease), toxicants (e.g., metals, volatile organics), and non-conventional pollutants (e.g., ammonia, nutrients). Some states have the authority to establish their own water-quality standards, which can be more stringent than federal standards established by the U.S. Environmental Protection Agency.
NON-CHEMICAL TECHNOLOGIES
The essential function of water treatment is to produce a change in water chemistry. More than 30 suppliers are known to provide non-chemical means of changing water chemistry, which can be grouped into five basic categories: magnetic, alternating-current (AC) induction, electrostatic, ultrasonic, and mechanical energy (cavitation).
Magnetic
Devices of this type (Figure 1) generally have one or more permanent magnets mounted to the outside or inside of a pipe. No other equipment or power is required. The orientation of the poles of the magnet(s) and the velocity of the water to be treated are critical to performance. Magnetic devices act through the Lorenz force (charged particles moving through a magnetic field), influencing ions in water to prevent scale. The process causes scale particles to precipitate within a water stream and be carried along with the water. Magnetic devices generally are not as effective in controlling bacteria and corrosion.
AC induction
These devices (Figure 2) consist of induction coils wrapped around a pipe or induction coils placed in cooling-water flow. AC is sent through the coils, imparting an induced electric field, along with variable magnetic fields, to water. Depending on the supplier, various frequencies, wave shapes, and strengths can be used. The generated current can be constant or pulsed. These devices are intended for scale, corrosion, and biological control.
Electrostatic
These devices (Figure 3) generate a powerful electrical field that affects the electrically charged dissolved ions of scale-causing minerals. Crystals grow (controlled precipitation) and remain suspended in the solution. Scale control is the main benefit of the technology; chemicals usually are required for corrosion and biological control.
Ultrasonic
In these devices (Figure 4), ultrasonic waves are generated using high-power, high-intensity ultrasonic transducers. As the sound waves pass though, water alternately is compressed and decompressed, with the potential for cavitation to result. Mostly, the devices are sold for microbiological control.
Mechanical energy
These devices (Figure 5) typically use pressure pumps to force water into plates, walls, or other water streams and produce cavitation. The cavitation forces soluble calcium carbonate (scale) to precipitate as small seed crystals that grow and are filtered from water. At the same time, dissolved carbon dioxide is removed, raising the pH of the water and inhibiting corrosion. Meanwhile, the effects of a high vacuum, cavitation, and high shear create an environment that destroys water-containing microorganisms without the possibility of resistance. Thus, these systems can be used for corrosion, scale, and microbiological control.
REDUCING WATER USE
One aspect of “greening” a cooling tower is improving its cycles of concentration (COC), or the number of times soluble mineral salts in water are concentrated. By increasing from two COC to four, savings in blowdown and makeup are approximately 67 percent and 33 percent, respectively (Figure 6). For a 300-ton cooling tower, this can translate to approximately 3.2 million gal. of water saved per year. If the current charge for fresh water were $1, and sewer charges were $2 per 1,000 gal., the total yearly savings would be $9,500.
In an actual cooling system, chemicals were eliminated, and the COC were increased from six to nine, resulting in annual savings of more than 5.4 million gal. of water. Moreover, use of the mechanical system has allowed more than 7 million gal. of non-potable water to be available for reuse on an annual basis.
RESPONSIBLE SPENDING
With some non-chemical treatment systems, a method of service comparable to a chemical treatment program is employed. This is effective because of the highly monitored, complex process of scale, corrosion, and bacteria treatment. The monthly service eliminates the capital expenditure of buying equipment.
Those non-chemical technologies can reduce cost by:
-
Increasing COC and reducing the cost of fresh water.
-
Decreasing the amount of water discharged to a POTW, reducing surcharges.
-
Eliminating regulated chemicals, allowing water to be discharged into receiving streams.
-
Allowing water reuse for non-potable applications.
-
Providing a multiyear fixed treatment cost (no swings in chemical pricing).
-
Providing rebates from utilities for water reuse.
REFERENCE
-
Washington State Department of Ecology. (2007). Cooling tower study: Facts & lessons learned. Retrieved from http://www.ecy.wa.gov/PROGRAMS/hwtr/TREE/exec_sum/ct.pdf
As technical director for VRTX Technologies, Philip Vella, PhD, directs all experiments and laboratory work pertaining to potential applications for controlled hydrodynamic cavitation. He holds a doctorate in inorganic chemistry from University at Albany, State University of New York. He can be contacted at [email protected].