Assessing the Role of HVAC Systems in Fighting COVID-19
By KENNETH M. ELOVITZ, P.E., and GARY M. ELOVITZ, P.E., Energy Economics, Inc., Newton Centre, MA
The primary role of HVAC systems is to maintain comfort, usually measured by temperature, humidity, and odor control in the conditioned space. Because many HVAC systems move air and some infectious diseases travel by air, there is interest in whether HVAC systems can play a role in managing infectious disease transmission.
HVAC systems cannot be the primary means of infectious disease control. However, increasing outdoor air provides dilution ventilation so can be a benefit. Also, better filtration (such as upgrading to MERV 13 filters) can help reduce the concentration of virus particles in the indoor environment by reducing the number of small particles (aerosols) that can harbor viral material.
Those measures address aerosols that can carry virus particles throughout a room. The HVAC system likely will not have much effect on close range transmission by the large droplets that people expel even when speaking but especially when coughing or sneezing.
While technological solutions like ultraviolet disinfection and bipolar ionization have been proposed as ways to reduce viral contamination in the conditioned space, there is not enough independent testing or practical experience to quantify their effect.
BACKGROUND
Viruses spread from person to person by one or more of three primary means:
- People touch contaminated surfaces and then touch their mucous membranes where the virus can enter the body. HVAC systems have little to no role in managing that type of transmission;
- Infected people release droplets that contain virus particles, and other people inhale those droplets. Large droplets (generally larger than 10 µm in diameter) tend to fall to the floor within 3 to 6 feet. However, large droplets can travel farther if propelled at high velocity, such as from a sneeze or cough. HVAC systems have little role in managing large droplets;
- Infected people also release virus particles in tiny droplets called aerosols (typically 5 µm and smaller) that behave like gases. In addition, moisture in large droplets evaporates quickly, so the remaining nuclei that potentially contain infectious virus particles can become small droplets or aerosols that remain in the air and travel on air currents.1 Because aerosols travel with air currents, HVAC systems might have a role in managing transmission by aerosols.
Canadian journalist Jonathan Kay analyzed 58 separate COVID-19 "super spreader" events in 28 countries. Kay's study covered the general public, not hospitals or nursing homes. Nineteen of the "super spreader" events occurred at parties or other "liquor-fueled" celebrations. Fourteen occurred at religious services or funerals. Six occurred at business meetings or networking events, raising some concern about exposure in the workplace, especially in small work groups where people are in close contact for extended periods. None occurred where people are in casual or incidental contact, such as while shopping. Except for three incidents at sports events (where people are in close proximity and likely talking or cheering loudly), all the events occurred indoors. That analysis led Kay to conclude that the biggest risk for COVID-19 transmission is direct, face-to-face, "ballistic delivery" of droplets.2
BECOMING INFECTED
Becoming infected requires an infectious dose of the virus. An infectious dose is the product of concentration and time. Some experts estimate that inhaling 1,000 infectious virus particles is enough for the COVID-19 virus to take hold. Studies based on influenza suggest that normal breathing (at rest) releases about 20 viral particles/minute. If a nearby person were to inhale all those particles (unlikely), the person would need 50 minutes of exposure to become infected. Speaking increases respiratory droplet production to about 200/minute. So a direct, face-to-face conversation could infect another person in five minutes (in the unlikely event that the recipient inhales all the exhaled particles from an infected person).3
The biggest danger for COVID-19 infection is close-range contact with infected people who are talking loudly or are otherwise exhaling heavily for at least several minutes. The HVAC system does not have much effect on those conditions. HVAC systems that provide outside air for ventilation and filtration can have a role in reducing airborne transmission of infectious disease by reducing the number of infectious particles in the room air.
A technique that relates infectious particle concentration, exposure time, and outside air ventilation might be a useful tool to guide analysis of the role HVAC systems can have in managing infectious aerosols. The Wells-Riley equation4 provides a method to predict the likelihood that a person will be infected by a virus:
Wells defined a quantum as the amount of infectious material required to infect 63.2% of the people in an enclosed space. 63.2% is (1 - 1/e) or the fraction infected in the first time constant of an exponential growth curve. The pulmonary ventilation rate for an adult is about 0.5 m3/hour or 0.29 CFM.5
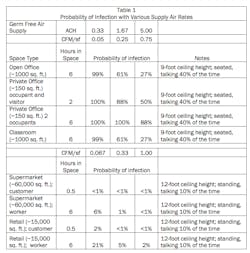
Table 1 applies the Wells-Riley equation to express the relative likelihood that an infected person releasing infectious aerosols will infect other people as a function of exposure time and outside air ventilation rates for several occupancy types.
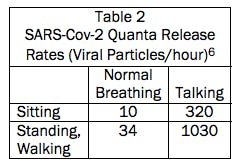
Table 2 lists the viral particle release rates used to generate Table 1.
The Wells-Riley equation and Table 1 do not say anything about the level of risk that might be acceptable. Their main value is to show the effect of changes in exposure time and ventilation on relative risk.
Table 1 suggests that no practical amount of ventilation can be relied on or expected to protect occupants over long exposure times like the 6 or 8 hours people might spend together in an office or school classroom. Similarly, ventilation is unlikely to succeed as the prime means of protection for people in close contact in a small space like a private office. However, short term, intermittent contact in large spaces like a trip to the supermarket is unlikely to spread infection.
Infected people release viral particles in a range of droplet sizes. Table 1 analyzes airborne spread of aerosols, so could overstate the viral load to the extent viral particles are in large droplets that do not travel with air currents. At the same time, Table 1 is based on a well mixed space. At least in the short term, the viral particle concentration will be higher near the infected person. That factor tends to make Table 1 understate the risk. Taken together, those two factors tend to balance, making Table 1 reasonable for evaluating relative benefits of changes in air supply. Ventilation can be part of an overall program to control the spread of infection by aerosols but is not a total solution to airborne infection control, especially if infection spreads by other means like large droplets.
That introduction provides a frame of reference for considering possible HVAC system modifications in an effort to manage COVID-19 spread by aerosols.
INCREASE OUTSIDE AIR
Outside air provides dilution ventilation for odors and other contaminants in the space, including virus aerosols. Every cubic foot of contaminant-free air introduced to the space pushes a cubic foot of air with its contaminants out of the space.
Table 1 shows relative risks at various levels of outside air per person. Depending on the system type and age (design practices change over time), outside air provided for ventilation might range from as little as 5 - 7 CFM/person in auditoriums and worship spaces to as much as 15 - 20 CFM/person in classrooms and offices. Table 1 expresses ventilation in terms of air changes/hour for the space. Depending on space size and occupant density, 5 CFM/person of outside air might provide between 0.33 and 1.5 air changes per hour of outside air.
Additional ventilation can dilute the particle concentration in the space. However, even if it were practical to deliver enough outside air to provide dilution equivalent to being outdoors, close range, large droplet transmission would remain a risk that the HVAC system cannot manage: "{S}tudies indicate that the interaction of coughed flow with high initial velocity with the free convection flow around the human body and the ventilation flow will be different than the flow of exhalation with much lower initial velocity (Gupta, et al. 2010), suggesting that the strategy of supplying extra amounts of outdoor air aiming to dilute the polluted room air may not be effective in protecting from airborne cross-infection due to coughing."7
Observe the following caution when considering increasing outside air: Outside air is generally warmer and more humid, or colder and drier, than the conditioned space. As a result, increasing minimum outside air quantities could increase cooling and heating loads. Adjustments to system capacity such as changing control settings or chilled/hot water flows might be needed to maintain desired space temperature and moisture levels. Be aware that with increased capacity to offset increased loads comes increased operating cost for cooling and heating energy.
ENHANCE FILTRATION
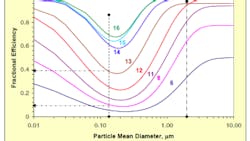
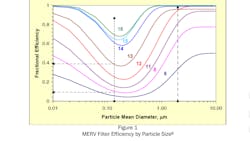
To the extent the coronavirus transmits from person to person by fine aerosols, improved filtration can reduce the risk of transmission by reducing the concentration of infectious particles in the air. Lower infective aerosol concentration effectively increases the amount of time required for recipients to inhale an infectious dose of virus particles. Figure 1 shows that MERV 13 filters are 95% efficient at removing those aerosol sized (2 µm) particles.
Notably, 2" pleated filters with a MERV 13 rating are readily available, so could be an easy retrofit in many commercial air handling units.
Particles in indoor air will be a mix of particles of various sizes. On average, MERV 13 filters might be 60% to 70% efficient at removing particles that contain viral material. At 67% (2/3) removal efficiency, 30 CFM of MERV 13 filtered, recirculated air works like 20 CFM of "germ free" air supply to a space when applying the Wells-Riley equation and the analysis in Table 1.
Enhanced filtration can contribute to the control of airborne infection that spreads by aerosols.
However, enhanced filtration cannot address short distance spread by large droplets so is not a standalone solution to airborne infection control.
Observe the following cautions when considering enhanced filtration:
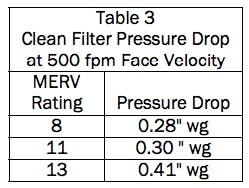
(1) Higher efficiency filters have higher pressure drop than the standard filters they replace. Table 3 lists one manufacturer's clean filter pressure drop at 500 fpm face velocity.
Higher pressure drop means reduced airflow, which reduces heating/cooling capacity and outside air supply (dilution ventilation to the space. But by how much? Each system needs an engineering evaluation, but take a 25-ton rooftop heating and air conditioning unit that serves 10,000 sq. ft. of commercial space as an example. The unit might be rated to deliver 10,000 CFM with 1.6" wg of static pressure available to overcome duct pressure drop (estimated 3.6" wg fan static pressure). 0.13" wg of added resistance to upgrade to MERV 13 filters would add less than 5% to system total pressure drop with a corresponding minimal reduction in airflow.
By contrast, a 3-ton water source heat pump or fan coil unit might be rated to deliver 1200 CFM at 0.25" wg external static pressure (0.75" wg fan static pressure). 0.13" wg of added resistance to upgrade to MERV 13 filters is almost 20% of system pressure drop so could cause a substantial drop in airlfow. A middle ground, such as MERV 11 filters with a tiny 0.02" wg increase in pressure drop over "standard" MERV 8 filters, might be considered for these small systems. If, as our research suggests, a reasonable goal is improvement as opposed to achievement, half a loaf might be better than none;
(2) Increasing fan speed to overcome the increased pressure drop and maintain original airflow will increase motor load, so check that the fan motor is not overloaded;
(3) Filters might have to be changed more frequently. Some MERV 13 filters achieve their efficiency with electrostatic coatings on the filter media. It is not clear how well the electrostatic coating maintains its effectiveness over time;
(4) Different practices and procedures (including personal protective equipment) might be needed when changing and disposing of filters believed to hold an accumulation of virus particles.
ULTRAVIOLET DISINFECTION
Ultraviolet radiation has been explored as a way to reduce the spread of airborne infections at least since the 1930s9. Ultraviolet radiation can disinfect surfaces, room air (called upper air systems), and air in ducts.10 To inactivate an organism (such as a virus), the ultraviolet radiation must strike the organism. Upper air (room) systems rely on air circulation in the room to carry microorganisms to the upper air zone where sufficient ultraviolet light shine on them.
Ultraviolet disinfection effectiveness is a function of dose, which is radiant intensity times time:
Although ultraviolet disinfection systems have some history and track record reducing mold and algae in air handling unit cooling coils and condensate pans, there is little definitive evidence of its effectiveness for controlling infectious particles in airstreams.11
One manufacturer reports that a dose of 1200 µw-s/cm2 effectively kills 99% of coronavirus organisms.12 Typical duct or air handling unit ultraviolet disinfection systems produce intensity in the range of 100 µw/cm2.13 Achieving a 1200 µw-s/cm2 dose would therefore require as much as 12 seconds exposure time, or an impractical 100 feet of duct if air flows only 500 feet/minute.
Higher intensity to reduce required duct length quickly increases electrical requirements with resulting waste heat gain and added cooling load. Reducing the dose reduces the kill rate. A workable balance between a practical installation and effective disinfection might be difficult to find.
Observe the following cautions when considering ultraviolet disinfection:
(1) Ultraviolet radiation can harm people. Ducts and air handling units might need safety measures like switches that automatically turn off the ultraviolet lights when access doors are opened. Upper air systems in spaces might need signage and local disconnect switches to protect workers;
(2) Do not overlook the electrical load for the ultraviolet disinfection system. Typical ultraviolet disinfection system output is 1/2 to 1/3 the lamp input watts. So a 50-watt UV lamp might deliver 15 to 25 watts of ultraviolet radiation. Additional circuits and wiring might be needed. The system will incur operating cost to run the UV lamps and add cooling load to offset waste heat.
BIPOLAR IONIZATION TREATMENT
Bipolar ionization breaks air into positive and negative ions. Those ions diffuse through the space and either react with or attach to particles. Reactions break down organic compounds and destroy odors. Attaching to particles causes them to agglomerate into larger particles that fall to the floor or are more readily filtered. Bipolar ionization systems are available for use in the occupied space or in air handling units. They are available from several manufacturers. There are no general design guides for engineers, so the manufacturers custom design each system. Observe the following cautions when considering ionization treatment:
(1) There are no industry standards or test protocols for bipolar ionization systems, so performance is not verified. There is some track record for reducing odors and destroying volatile organic compounds, but solid data on killing viruses is scarce;
(2) Some ionization systems produce ozone as a byproduct. Ozone is a disinfectant, but it is also an irritant. Ozone attacks rubber so could deteriorate belts, gaskets, and seals in air handling units;
(3) During a Q&A session at the 2020 ASHRAE Virtual Conference, indoor air quality expert Max Sherman of Lawrence Berkeley Laboratories was hesitant about this technology. He reasoned that improving indoor air quality usually involves removing contaminants from the air. He was uneasy about the effect of adding something to the air, especially when long term effects on people are not known.
SUMMARY AND CONCLUSION
HVAC systems have little role to play in controlling the spread of infectious disease that occurs by contact with infected surfaces and close range injection of large droplets (coughing, sneezing, or even loud talking or singing). To the extent that a disease spreads by small aerosols that travel with indoor air currents, HVAC systems might be able to provide some risk reduction by increasing outside air for ventilation or enhancing filter efficiency. Upper air/in room ultraviolet disinfection can be effective, but the value of UV disinfection for viruses in air in ducts is less certain. There is no solid evidence that HVAC systems can be a primary means of control or that HVAC systems are a primary conduit for spreading infectious disease.
############
A member of HPAC Engineering’s editorial advisory board, author Ken Elovitz is both a mechanical and electrical engineer, as well as in-house counsel for Energy Economics, Inc., a consulting firm based near Boston MA. Email him at [email protected].
Co-author Gary Elovitz, like Ken, also has been with Energy Economics since 1982. He is an engineering consultant experienced in design, analysis, and troubleshooting of HVAC and power distribution systems. Email him at [email protected].
###########
1 Xie, X., Li, Y., Chwang, A. T. Y., Ho, P. L., & Seto, W. H. (2007). How far droplets can move in indoor environments–revisiting the Wells evaporation–falling curve. Indoor air, 17(3), 211-225;
2 Kay, J. (2020). COVID-19 superspreader events in 28 countries: Critical patterns and lessons. Quillette, April, 23;
3 https://www.erinbromage.com/post/therisks-know-them-avoid-them, May 6, 2020. Erin Bromage is an Associate Professor of Biology at the University of Massachusetts Dartmouth;
4 Noakes, C. J., Beggs, C. B., Sleigh, P. A., & Kerr, K. G. (2006). Modelling the transmission of airborne infections in enclosed spaces. Epidemiology & Infection, 134(5), 1082-1091;
5 Ibid.
6 Buonanno, G., Stabile, L., & Morawska, L. (2020). Estimation of airborne viral emission: quanta emission rate of SARSCoV-2 for infection risk assessment. https://www.medrxiv.org/content/10.1101/2020.04.12.20062828v1;
7 Memarzadeh, F., & Xu, W. (2012, March). Role of air changes per hour (ACH) in possible transmission of airborne infections. In Building Simulation (Vol. 5, No. 1, pp. 15-28). Tsinghua Press;
8 Kowalski, W. J., & Bahnfleth, W. P. (2002). MERV filter models for aerobiological applications. Air Media, Summer, 1;
9 Wells, W. F., Wells, M. W., & Wilder, T. S. (1942). The Environmental Control of Epidemic Contagion. I. An Epidemiologic Study of Radiant Disinfection of Air in Day Schools. American Journal of Hygiene, 35(1), 97-121;
10 Owen, M. S. (Ed.). (2019). 2019 ASHRAE Handbook: Heating, Ventilating, and Air-conditioning Applications: InchPound Edition, Chapter 62;
11 Memarzadeh, F., Olmsted, R. N., & Bartley, J. M. (2010). Applications of ultraviolet germicidal irradiation disinfection in health care facilities: effective adjunct, but not stand-alone technology. American Journal of Infection Control, 38(5), S13-S24;
12 https://www.uvdi.com/wpcontent/uploads/2020/03/UVC-AirstreamFlyer_MKTFM-455-Rev-A.pdf, retrieved 7/16/20;
13 Ibid. and Kowalski, W. J., & Bahnfleth, W. P. (2000). UVGI design basics for air and surface disinfection. HPAC Engineering, 72(1).
About the Author
Ken Elovitz, P.E., Esq.
A longtime member of HPAC Engineering's Editorial Advisory Board, Kenneth M. Elovitz, P.E., Esq., is an engineer (mechanical and electrical) and in-house counsel for Energy Economics, Inc., a consulting engineering firm in Foxboro, MA. He can be reached at [email protected].